Reverse Engineering the Fruit Fly Brain
Our research focuses on Drosophila as an attractive model system for understanding the logic of olfactory perception because a fruit fly (i) exhibits complex olfactory-driven behaviors, (ii) has a nervous system that is numerically five orders of magnitude smaller than that of vertebrates (the fly brain contains approximately 100,000 neurons) and, (iii) makes it possible to use an extensive genetic toolbox to visualize and alter neural circuits in the olfactory system. One additional factor that makes Drosophila so attractive for olfactory research is (iv) the uniquely comprehensive description of its sensory periphery, including a complete molecular characterization of olfactory sensory neurons and their projections into the brain. All of these factors make Drosophila a powerful system for analyzing the neural circuit basis of behavior and provide an unmatched platform for investigating the logic of olfactory information processing in the brain.
Biological Validation of Neural Circuit Models of the Fruit Fly Brain
Efforts at reverse engineering the brain must ultimately confront the need to validate hypotheses regarding neural information processing against actual biological systems. In order to achieve biological validation of the Neurokernel, the computational modeling of the fly brain must be tightly integrated with increasingly precise electrophysiological techniques and the recorded data evaluated with novel system identification methods. This will enable direct comparison of the output of models executed by Neurokernel to that of corresponding neurons in the brain regions of interest.
The methodology for the biological validation of Neurokernel circuits and architectures is schematically depicted below.
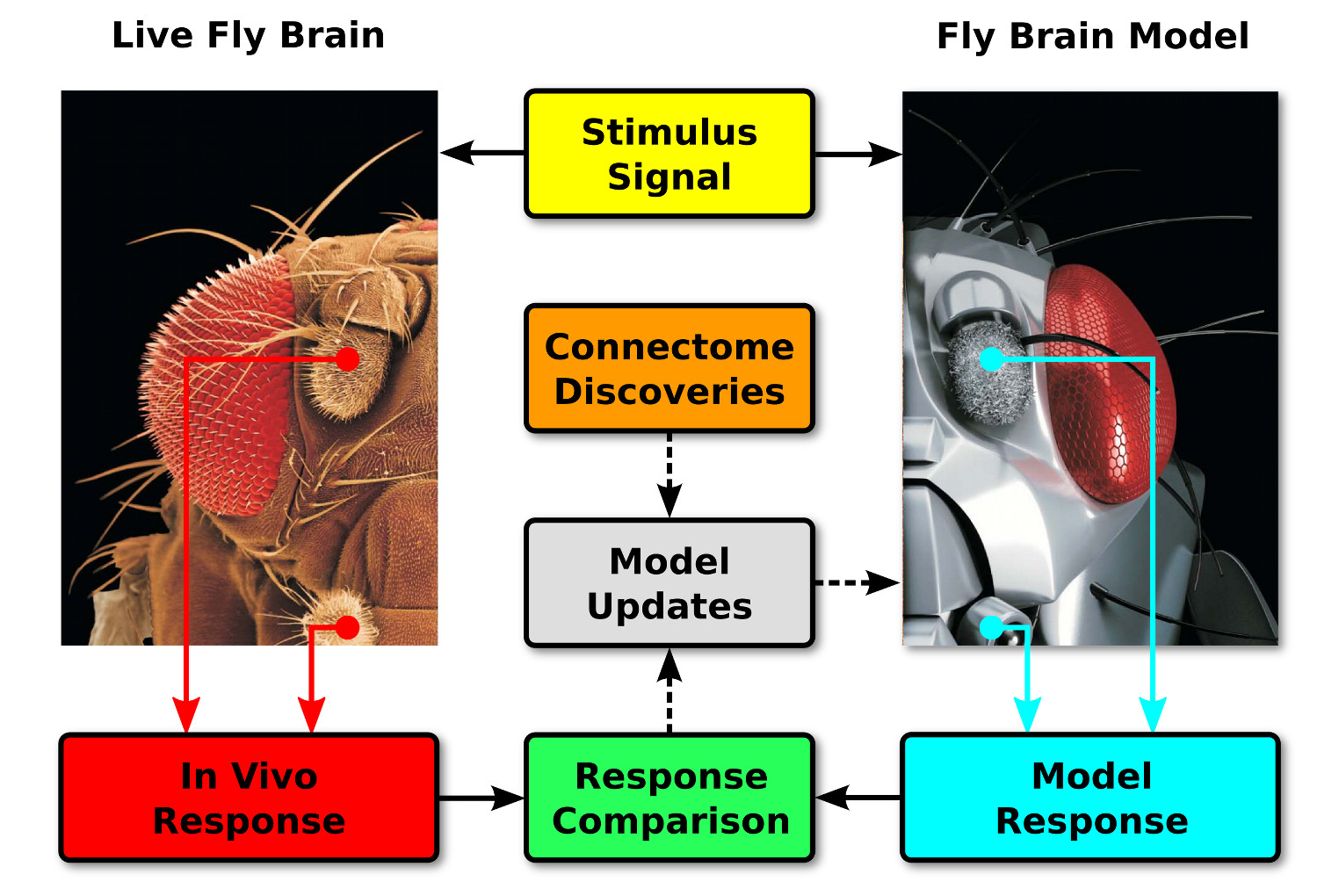
Neural responses to sensory stimuli are recorded from the live fly brain in real time and compared to the computed responses of the corresponding components in a fly brain model executed on the same time scale. Discrepancies between these responses and new connectome data may be used to improve the model's accuracy
Reverse Engineering the Early Olfactory System of the Drosophila
We believe that the lack of a deeper understanding of how Olfactory Sensory Neurons (OSNs) encode odors has fundamentally hindered progress in understanding olfactory signal processing in higher brain centers. Moreover, the lack of precise stimulus delivery and measurement systems has fundamentally limited the progress of system identification in olfaction.
To address this limitation, we developed a novel in vivo experimental setup with precise and reproducible delivery of airborne stimuli. This experimental setup enabled us to apply system identification methods to OSNs in Drosophila. Currently, we are applying time-varying odor stimuli and are recording in vivo the response of Projection Neurons (PNs) postsynaptic to OSNs. These novel recordings have shown that individual OSNs and PNs encode the gradient and acceleration of odor concentration waveforms, respectively. This research was performed in collaboration with Dr. Richard Axel in the Axel Laboratory.
- Anmo J. Kim, Aurel A. Lazar and Yevgeniy B. Slutskiy, System Identification of Drosophila Olfactory Sensory Neurons , Journal of Computational Neuroscience, Vol. 30, No.1, February 2011, pp. 143-161, Special Issue on Methods of Information Theory.
- A.J. Kim, A.A. Lazar, and Y.B. Slutskiy, Projection Neurons in Drosophila Antennal Lobes Signal the Acceleration of Odor Concentrations, eLife 2015;10.7554/eLife.06651, June 2015.
- A.A. Lazar and C.-H. Yeh, Functional Identification of an Antennal Lobe DM4 Projection Neuron of the Fruit Fly, Computational Neuroscience Meeting, Volume 15, July 2014, Québec City, Canada.
Local Processing Units (LPUs) of the Early Vision System of the Fruit Fly
The retina and the lamina neuropil of the early visual system of the fruit fly brain are, respectively, described by the RFCs below. Open source code and documentation is available at http://neurokernel.github.io/docs.html.
- Aurel A. Lazar, Konstantinos Psychas, Nikul H. Ukani and Yiyin Zhou, A Parallel Processing Model of the Drosophila Retina, Neurokernel Request for Comments, Neurokernel RFC #3, August 2015.
- Aurel A. Lazar, Nikul H. Ukani and Yiyin Zhou, The Cartridge: A Canonical Neural Circuit Abstraction of the Lamina Neuropil - Construction and Composition Rules, Neurokernel Request for Comments, Neurokernel RFC #2, January 2014.
The above video shows a visual evaluation of the fruit fly retina as a parallel information pre-processor. The light intensity of the original scene is shown on the top left. The red circle indicates the visual aperture of the eye. The logarithm of the original scene is shown on the top right. On the bottom left, “Visual Aperture” shows the projection of the area inside the red circle in the original scene onto the hemispherical visual field. “Input to R1” shows the rate of photons arriving at the R1 photoreceptors. “Delayed by 40 [ms]” shows the “Input to R1” delayed by 40 [ms]. “log(Visual Aperture)”, “log(Input)” and “log(Delayed by 40 [ms])” show the log of the plot in, respectively, “Visual Aperture”, “Input to R1” and “Delayed by 40 [ms]”. Note that “Input to R1” is the actual input to the photoreceptors. On the bottom right, the responses of photoreceptors R1-R6 are shown, respectively, in each block. All hemispheres are viewed orthogonal to their base plane. As a result, they appear to be circular.
The Bionet Group is supported by grants from
 |
 |
 |
 |